InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the “Global Residual DNA Testing Market Size, Share & Trends Analysis Report Offering (Product {Consumables, and Instruments & Software}, and Services), by Test Type (Final Product Release Testing, Bulk Lot Release Testing, and Raw Material Testing), By Technology (Polymerase Chain Reaction, Threshold Assay, DNA Probe Hybridization, and Other Technologies), By Application (Monoclonal Antibodies, Cell & Gene Therapy, Vaccines, And Other Applications), By End-User (Pharmaceutical & Biotechnology Companies, CROS & CDMOS, And Academic & Research Institutes)-Market Outlook And Industry Analysis 2034″
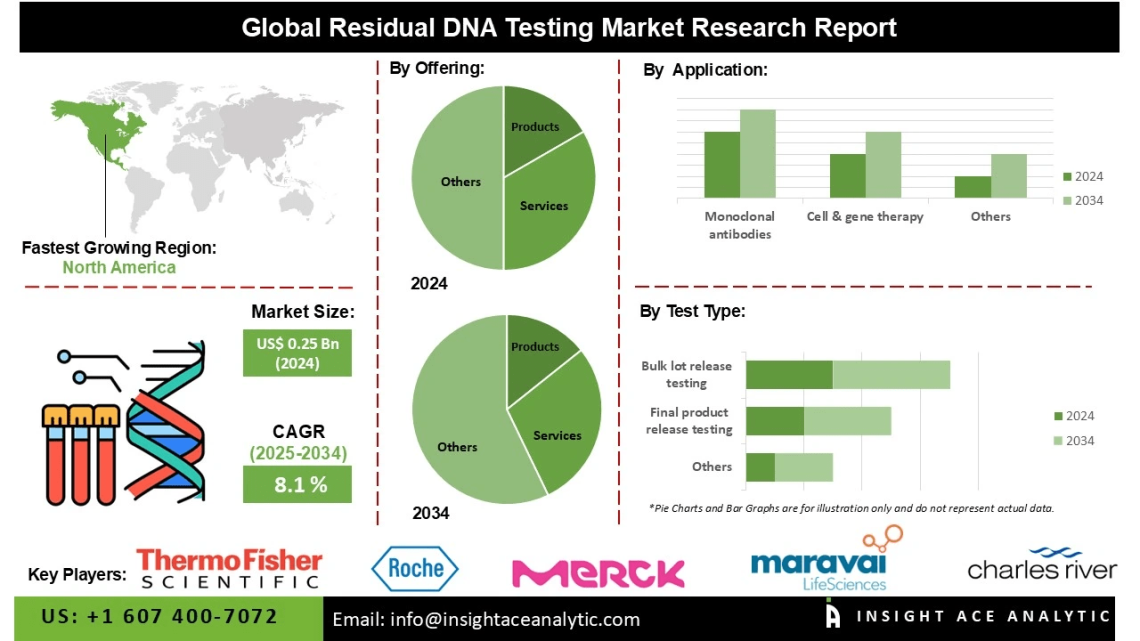
The Global Residual DNA Testing Market is valued at US$ 0.25 Bn in 2024 and is expected to reach US$ 0.54 Bn by the year 2034, with a CAGR of 8.1% during the forecast period of 2025-2034.
Get Free Access to Demo Report, Excel Pivot and ToC: https://www.insightaceanalytic.com/request-sample/3191
Residual DNA testing is a critical analytical technique employed to detect and quantify trace levels of host cell DNA in biopharmaceutical products following manufacturing. This testing ensures product safety, purity, and compliance with regulatory standards by confirming minimal residual contamination from production cells. The market for residual DNA testing is being driven by increased research and development investments, as biopharmaceutical and biotechnology companies prioritize product safety, regulatory adherence, and innovation.
Advanced biologics, vaccines, and gene therapies necessitate rigorous residual DNA assessment in accordance with guidelines from regulatory authorities such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Enhanced R&D funding facilitates the adoption of sophisticated analytical methods, including quantitative PCR (qPCR), digital droplet PCR (ddPCR), and next-generation sequencing, which provide superior sensitivity, accuracy, and throughput. Collaborative initiatives between research institutions and industry players further accelerate product approvals and contribute to market expansion.
The rapid growth of cell and gene therapies represents a significant driver for the residual DNA testing market. These therapies utilize living cells and genetic material, where stringent quality control is critical to mitigate risks such as immunogenicity or oncogenicity. As regulatory authorities increasingly mandate comprehensive residual DNA analysis for therapies targeting cancer, rare diseases, and genetic disorders, the adoption of high-throughput analytical technologies is expanding. The deployment of advanced techniques, including qPCR and next-generation sequencing, supports compliance, ensures safety and efficacy, and expedites product development, thereby propelling market growth.
List of Prominent Players in the Residual DNA Testing Market:
- Thermo Fisher Scientific Inc. (US)
- Merck KGaA (Germany)
- Eurofins Scientific (Luxembourg)
- Charles River Laboratories (US)
- Bio-Rad Laboratories, Inc. (US)
- QIAGEN (Germany)
- Hoffmann-La Roche Ltd (Switzerland)
- Maravai LifeSciences (US)
- FUJIFILM Corporation (Japan)
- SGS Société Générale de Surveillance SA. (Switzerland)
- Jiangsu Hillgene Biopharma Co., Ltd (China)
- Revvity (US)
- Intertek Group plc (UK)
- WuXi AppTec (China)
- Sartorius AG (Germany)
- Lonza (Switzerland)
Read Overview Report- https://www.insightaceanalytic.com/report/residual-dna-testing-market/3191
Market Dynamics:
Drivers:
The residual DNA testing market is experiencing robust growth, propelled by the increasing approvals and expanding pipelines of biologics and biosimilars. Regulatory authorities, including the FDA and EMA, are enforcing stricter safety and quality standards, requiring manufacturers to ensure minimal residual host-cell DNA in therapeutic products to mitigate safety risks and preserve efficacy. The growing volume of monoclonal antibodies, vaccines, and recombinant proteins entering clinical and commercial stages significantly drives the demand for sensitive and reliable DNA testing solutions. Furthermore, the rapid expansion of the biosimilars sector, driven by patent expirations of leading biologics, sustains demand for rigorous residual DNA testing to maintain regulatory compliance and safeguard patient safety.
Challenges:
High costs associated with advanced residual DNA testing techniques remain a key restraint on market growth. Sophisticated methodologies such as qPCR, next-generation sequencing (NGS), and capillary electrophoresis require substantial investment in specialized instruments, reagents, and skilled personnel, leading to elevated operational expenses. Smaller biotech companies, academic laboratories, and firms in developing regions often face financial limitations that hinder adoption. In addition, ongoing requirements for calibration, validation, and strict regulatory compliance further increase costs. These financial and operational barriers limit wider adoption of residual DNA testing, concentrating usage among well-capitalized pharmaceutical and biotechnology organizations.
Regional Trends:
In North America, the residual DNA testing market is expanding rapidly due to strong adoption of biopharmaceuticals and rigorous regulatory oversight from agencies such as the FDA and Health Canada. The increasing production of biologics, vaccines, and gene therapies necessitates precise monitoring of residual host-cell DNA to ensure product safety. Investments in next-generation biomanufacturing facilities and growing outsourcing to contract research and testing organizations are further stimulating demand for high-sensitivity DNA testing technologies.
Europe’s market growth is similarly driven by stringent regulatory frameworks emphasizing safety in biopharmaceuticals, vaccines, and cell-based therapies. Regulatory bodies, including the EMA, mandate rigorous residual DNA testing to maintain biologics within safe limits, fostering the adoption of advanced testing methodologies. Additionally, rising production of biologics, expansion of cell and gene therapy research, and increased outsourcing of testing services to specialized laboratories are contributing to the growth of the European residual DNA testing market.
Unlock Your GTM Strategy: https://www.insightaceanalytic.com/customization/3191
Recent Developments:
- April 2025: Thermo Fisher Scientific, Inc. declared the opening of its Advanced Therapies Collaboration Centre (ATXCC) in California, US. This facility is designed to accelerate the development and commercialization of cell therapies.
Segmentation of Residual DNA Testing Market-
By Offering-
- Product {Consumables, and Instruments & Software}
- Services
By Test Type-
- Final Product Release Testing
- Bulk Lot Release Testing
- Raw Material Testing
By Technology-
- Polymerase Chain Reaction
- Threshold Assay
- DNA Probe Hybridization
- Other Technologies
By Application-
- Monoclonal Antibodies
- Cell & Gene Therapy
- Vaccines
- Other Applications
By End-User-
- Pharmaceutical & Biotechnology Companies
- CROS & CDMOS
- Academic & Research Institutes
By Region-
North America-
- The US
- Canada
Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
Middle East & Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
About Us:
InsightAce Analytic is a market research and consulting firm that enables clients to make strategic decisions. Our qualitative and quantitative market intelligence solutions inform the need for market and competitive intelligence to expand businesses. We help clients gain competitive advantage by identifying untapped markets, exploring new and competing technologies, segmenting potential markets and repositioning products. Our expertise is in providing syndicated and custom market intelligence reports with an in-depth analysis with key market insights in a timely and cost-effective manner.
Contact us:
InsightAce Analytic Pvt. Ltd.
Visit: https://www.insightaceanalytic.com/
Tel : +1 607 400-7072
Asia: +91 79 72967118
info@insightaceanalytic.com